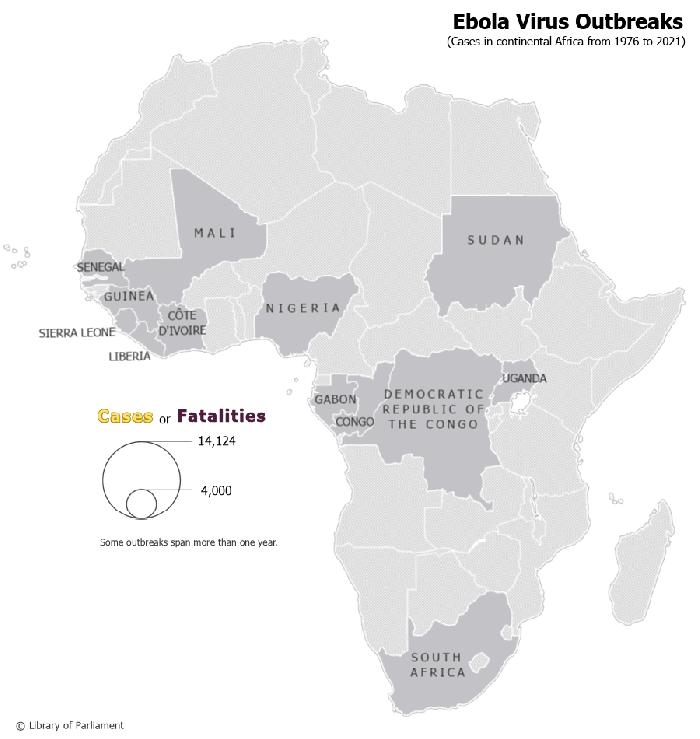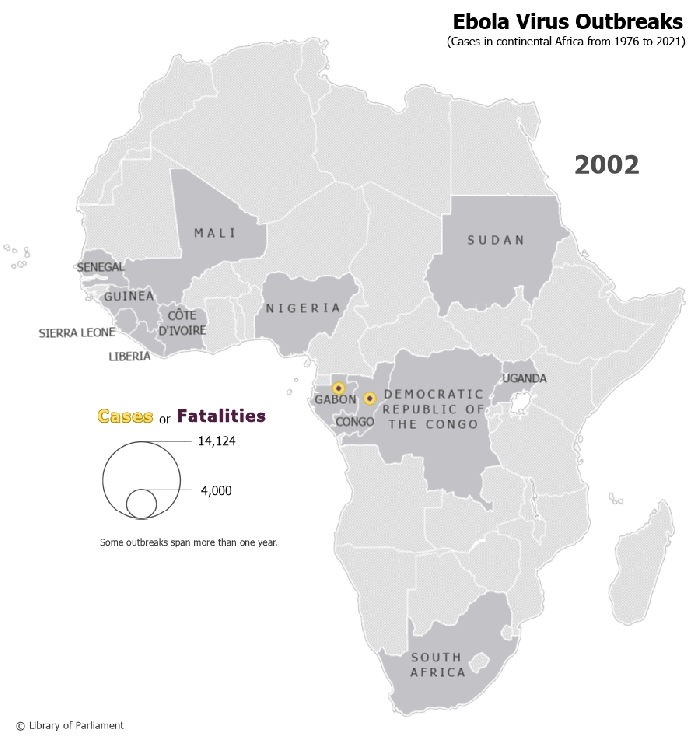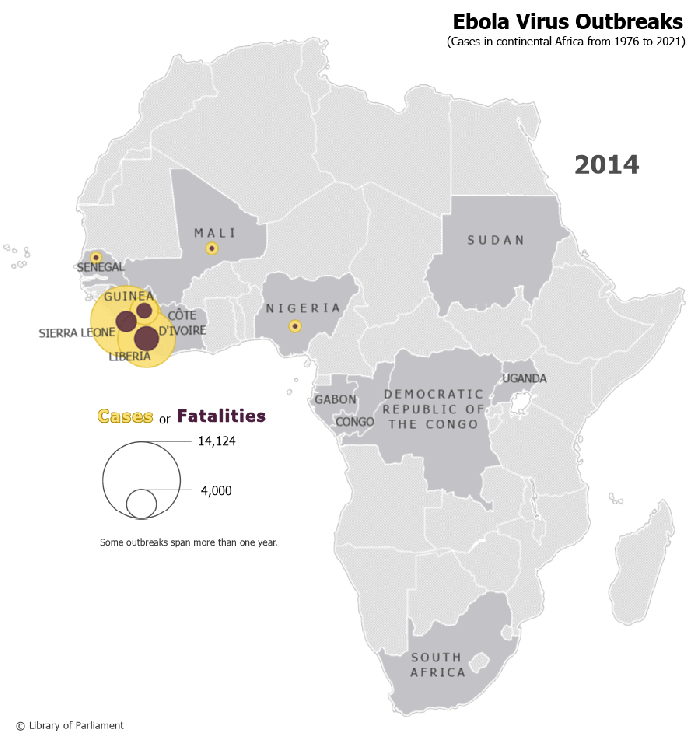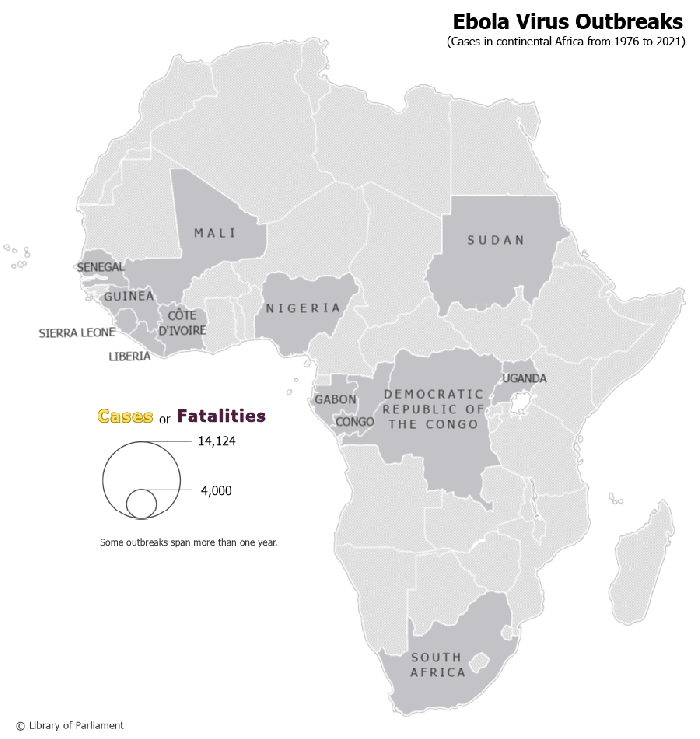
The 2014–2016 Ebola virus outbreak in West Africa was the largest in history, resulting in more cases and deaths than all other Ebola outbreaks combined. The virus infected 28,610 people, primarily in Guinea, Liberia and Sierra Leone, leading to 11,308 deaths. Since then, several new Ebola outbreaks have occurred, including one in the Democratic Republic of the Congo (DRC) between 2018 and 2020. That outbreak, the second largest ever, resulted in 3,481 cases and 2,299 deaths.
Figure 1 – Ebola Virus Outbreaks in Continental Africa, 1976–2021
Map prepared by the Library of Parliament, Ottawa, 2021, using data from Natural Earth, 1:50m Cultural Vectors, version 4.1.0; World Health Organization, “Table: Chronology of previous Ebola virus disease outbreaks,” Ebola virus disease; and Centers for Disease Control and Prevention, “Ebola Virus Outbreaks by Species and Size, Since 1976,” Ebola (Ebola Virus Disease). The following software was used: Esri, ArcGIS Pro, version 2.8.3.
The West African outbreak led to renewed urgency to approve and deploy an Ebola vaccine. It also highlighted the need for more effective public health measures to contain cross-border infection. However, despite several expert reports produced following the outbreak, the implementation of key reforms to the global health architecture has been inconsistent, which may have affected preparedness for the COVID-19 pandemic.
What is Ebola?
Four Ebola virus species, referred to collectively as the Ebola virus, cause the rare and often fatal disease in humans called Ebola virus disease (EVD). It was first identified in 1976 in two outbreaks that occurred simultaneously in what are now South Sudan and the DRC.
The Ebola virus is contracted by, or transmitted to, humans through direct contact with the infected bodily fluids of animals or humans. The virus, which disrupts blood clotting and can result in internal bleeding, has an average fatality rate of 50%. Although the Ebola virus does not appear to mutate very often, one 2016 study found that the magnitude of the West African outbreak enabled “an unprecedented number of viral mutations,” which may have heightened the infectiousness of the virus.
The West African Outbreak
The initial case that triggered the West African outbreak is believed to have been that of a young boy from rural Guinea who was infected by bats. By mid-2014, the virus had spread across Guinea and into Liberia and Sierra Leone. Imported cases were also identified in other African countries and in Italy, Spain, the United Kingdom and the United States. In August 2014, the World Health Organization (WHO) declared the deteriorating situation in West Africa a Public Health Emergency of International Concern, and called for a coordinated global response.
Local and international health officials worked to contain the spread through contact tracing, testing, treatment, quarantine, safe burial practices and health education. As part of the Canadian response, Canadian Armed Forces medical and military personnel were deployed to Sierra Leone to provide medical care and strategic support. The Public Health Agency of Canada (PHAC) also sent experts to the region to provide laboratory, emergency management and diagnostic support.
In an effort to prevent cross-border transmission, many countries imposed restrictions on travellers from West Africa and established enhanced screening procedures at airports. For example, relying on section 58 of the Quarantine Act, the Canadian government made orders in council that allowed quarantine officers at Canadian borders and ports of entry to question, assess and, if necessary, detain travellers entering Canada from affected countries.
By June 2016, Guinea, Liberia and Sierra Leone were all declared EVD-free, but the virus had exacted a heavy human and socio-economic toll. It disrupted economic activity in several sectors, including agriculture, mining, manufacturing and transportation; it also disturbed cross-border trade. According to the World Bank, the total estimated gross domestic product (GDP) loss for Guinea, Liberia and Sierra Leone was US$2.8 billion, which represents an average GDP loss of US$125 per person in the three countries.
A 2018 academic article argued that many estimates of the economic burden of the outbreak do not include its social costs. According to the article, the estimated comprehensive economic and social costs were US$53.19 billion globally. This includes, as the most significant cost, US$18.78 billion due to deaths from non-EVD causes. These deaths were related to reduced health care services stemming from the loss of health care workers and diversion of health resources. In the case of Liberia, 8% of the country’s doctors, nurses and midwives died of EVD. Given their role as the primary caregivers within the home and as front-line health workers, women were disproportionally more likely than men to be infected by Ebola during the outbreak.
Vaccine Development and Use
Canada played a significant role in the development of an Ebola vaccine. In 2001, scientists from PHAC’s National Microbiology Laboratory began work on a vaccine. In October 2014, the Government of Canada sent 800 vials of its Ebola vaccine candidate to the WHO to help contain the outbreak in West Africa.
The vaccine candidate was licensed to NewLink Genetics and, in 2014, the company announced its collaboration with Merck to conduct clinical trials with a view to manufacturing and distributing the vaccine. In November 2019, the European Commission approved the Ebola vaccine – commercialized as Ervebo – and the vaccine was prequalified for use by the WHO. In July 2020, a second Ebola vaccine – developed by Janssen – was approved by the European Commission.
During the 2018–2020 Ebola outbreak in the DRC, approximately 300,000 people received the Ervebo vaccine as part of a containment approach called “ring vaccination.” Under this approach, the primary and secondary contacts of infected persons, as well as health care and frontline workers, are vaccinated to create a “ring” of immunity intended to disrupt the chain of transmission. Despite the vaccine, controlling that outbreak was particularly challenging because it took place in an active conflict zone. Dozens of armed groups operate in eastern DRC, which complicated efforts to reach and treat those affected or at risk of infection.
In January 2021, a global emergency stockpile of 500,000 Ervebo vaccines was established with financial support from Gavi, the Vaccine Alliance. Vaccines from that stockpile were used to combat outbreaks that began in the DRC and Guinea in February 2021. The outbreak in the DRC resulted in 12 cases and 6 deaths and was declared over in May 2021, while the outbreak in Guinea led to 23 cases and 12 deaths and ended in June 2021.
Key Lessons
In November 2015, the Independent Panel on the Global Response to Ebola released a report into the international response to the West African outbreak. The panel criticized the WHO for its failure to quickly mobilize international assistance and for its “weak” in-country technical capacity. Among its recommendations, it called on the WHO to strengthen its early warning capacity and to establish a dedicated centre for outbreak response.
This report, together with that of another independent body of experts and one by a high-level United Nations panel, prompted the WHO to establish a new health emergencies program in 2016. The WHO created the program to accelerate its emergency work and to provide more predictable financial, logistical and other support to countries and communities in need. However, groups of experts stated that the reforms enacted by the WHO in the aftermath of the West African outbreak “fail to achieve the ‘evolution’ in global health governance needed [to adequately protect society against] modern public health emergencies.”
The COVID-19 pandemic brought the WHO back under international scrutiny. Criticism of the WHO’s emergency response prompted the World Health Assembly (WHA) – the governing body of the WHO – to call for an independent evaluation. In May 2021, the Independent Panel for Pandemic Preparedness and Response referred to “gaps and failings at every critical juncture of preparedness for, and response to, COVID-19.” Among its recommendations, the panel called on WHO member states to strengthen the independence, authority and financing of the WHO, and on the WHO to develop a new rapid global alert system for outbreaks. WHO member states agreed to establish a working group to examine the panel’s findings and to create concrete proposals for the WHA to consider in 2022.
Additional Resources
Daniel G. Bausch, “The need for a new strategy for Ebola vaccination,” Nature Medicine, Vol. 27, April 2021, pp. 580–581.
European Medicines Agency, Ervebo (Ebola Zaire Vaccine [rVSV∆G-ZEBOV-GP, live]).
Claire Felter and Danielle Renwick, What is the Ebola Virus? Backgrounder, Council on Foreign Relations, 1 July 2020.
Megha Satyanarayana, “Vaccines could make big Ebola outbreaks a thing of the past,” Chemical & Engineering News (C&EN), 20 June 2021.
World Health Organization, Ebola virus disease.
Authors: Raphaëlle Deraspe and Brian Hermon, Library of Parliament
Categories: Health and safety, International affairs and defence